Formic acid is the active ingredient in MAQS® and Formic Pro®. In this post, we are going to explore some of the properties of formic acid that, when combined with NOD’s slow-release technology, make it a great acaracide.
From Ants to Birds to Bees: A Brief History of Formic Acid
Formic acid is an organic acid.“Organic” in chemistry means that a substance contains carbon. The reason we associate these types of compounds with organic (living) things is that the bodies of all living organisms are made up of carbon-containing molecules.
Formic acid is naturally produced and used by a huge range of living things — most famously, by ants. Indeed, the word formic comes from the latin formica, meaning ant, because this is where the compound was first identified. Ants spray formic acid from their abdomens to defend their colonies against attack.
Among the various uses of formic acid found in nature is one intriguing bird behaviour known as “anting.” Anting birds deliberately provoke ants by disturbing their nests, even picking up the insects and placing them into their feathers. Birds often endure numerous ant bites for the opportunity to bathe in formic acid released by the defensive ants. Scientists suspect that, among other things, the formic acid released by the ants helps birds rid themselves of parasites, including mites.

Alfred, an adult male Carrion Crow (Corvus corone), anting at the Jardin des Plantes in Paris.
PHOTO CREDIT: Marie-Lan Tay Pamart, Wikimedia Commons (licensed under Creative Commons Attribution 4.0 International).
Many organisms have put formic acid to work in many ways. Humans use formic acid in the production of sileage for cattle feed, as an energy source in fuel cells, as part of the of the leather tanning process – and of course, to treat honey bee colonies infested with Varroa destructor mites..
The Advantages of Evaporation
Formic acid is volatile. Although volatility might make us think of something unstable, in chemistry, a volatile substance is one that evaporates easily. Chemists use a measurement called vapour pressure to establish how volatile a substance is. Briefly, vapour pressure is the pressure created due to evaporation of a liquid that is sealed inside a closed container. The higher the vapour pressure, the more readily a substance evaporates, and consequently, the higher the pressure of the gas due to evaporation inside the container.
Vapour pressure, and hence volatility, depends on two things: the molecular properties of a substance, and the temperature. The vapour pressure of any substance increases as the temperature increases. For example, the vapour pressure of water at 25 °C is lower than that of formic acid at 25 °C. But the vapour pressure of both water and formic acid increases as the temperature increases. To put it simply, as it gets hotter, the rate of evaporation increases for any liquid.
Evaporation is key to the effectiveness of formic acid for varroa control, because gasses move around inside the colony much more readily than liquids or solids. But as we’ll see, not all gasses are created equally.
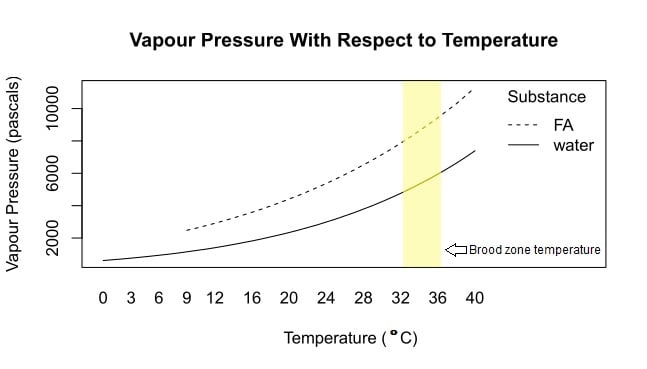
Vapour pressure is a measure of how easily a substance evaporates and is a function of the chemical properties of a substance along with the temperature. The higher the vapour pressure, the more readily a substance evaporates. Formic acid (FA) has a higher vapour pressure than water, hence formic acid is more volatile than water. All liquids become solid at some temperature, at which point evaporation ceases. Pure formic acid becomes solid at 8.3 C, whereas water becomes solid at 0 C, corresponding to the lower ends of the vapour pressure curves on this graph. Interestingly, 85% formic acid freezes at – 20 C while 75% formic acid freezes at – 30 C.Data generated by the Vienna Institute of Technology, Institute of Applied Physics Vapour Pressure Calculator.
Small Molecules Disperse More Easily
When strips of Formic Pro or MAQS are placed inside a hive, an invisible cloud of formic acid vapour begins to evaporate off the liquid formic acid that is suspended in the solid matrix of the strips. The cloud is made up of molecules of formic acid that constantly zoom around, bumping into each other and everything else.
All the zooming and bumping means that formic acid molecules tend to move away from areas of high concentration (e.g., where they evaporate out of the Formic Pro strip) toward areas where there is less formic acid. This process is called diffusion. Diffusion is the main force driving formic acid molecules into all the nooks and crannies of the hive – including into the sealed brood cells.

A brood cell cap seen under a dissecting microscope. The waxy outer layer has been scraped back to reveal the silk layer underneath. Unlike cappings over honey, which are air tight, brood cell cappings are porous, allowing gasses to move in and out of the cells. PHOTO CREDIT: Heather Broccard-Bell.
The rate of diffusion of any substance is related to the speed at which individual particles move around. The velocity of particles depends on temperature, how tightly the particles are packed together (density), and the size of the particles. When the temperature and density are the same, smaller molecules move faster than larger molecules. Formic acid is the simplest organic acid, made up of just two oxygen atoms, two hydrogen atoms, and one carbon atom. Compared to most other organic compounds, formic acid molecules are tiny. Hence, they can move around very efficiently to reach more areas within the hive.

Relative sizes of some familiar organic compounds along with their molar masses. Black are carbon atoms, white are oxygen atoms, and red are hydrogen atoms. IMAGE CREDIT: Formic Acid: Ben Mills, Wikimedia Commons (public domain), Oxalic Acid: Jynto, Wikimedia Commons (licensed under Creative Commons Zero, Public Domain Dedication.), Thymol: itineranttrader, Wikimedia Commons (public domain).
It’s All About Dose
Formic acid is naturally present in honey bee colonies, and is a normal component of honey. At these low levels, it is not harmful to humans, bees, or varroa mites. But raise the levels of formic acid enough, and it’s a different story1.
Studies have shown that formic acid levels need to reach about 20 ppm inside the colony to begin to kill varroa. NOD’s own studies have shown that levels of formic acid around 40 – 80 ppm over several days are ideal to achieving varroa control. Therefore, the key to effective treatment depends on getting the dose just right.
Although beekeepers have used liquid formic acid to control varroa since the 1970s, the results have sometimes been inconsistent. The main issue is that the rate of evaporation of liquid formic acid can be difficult to control under varying temperature conditions. The development of MAQS and Formic Pro with their slow-release technology has made it much easier for beekeepers to ensure that colonies get the proper dose2.
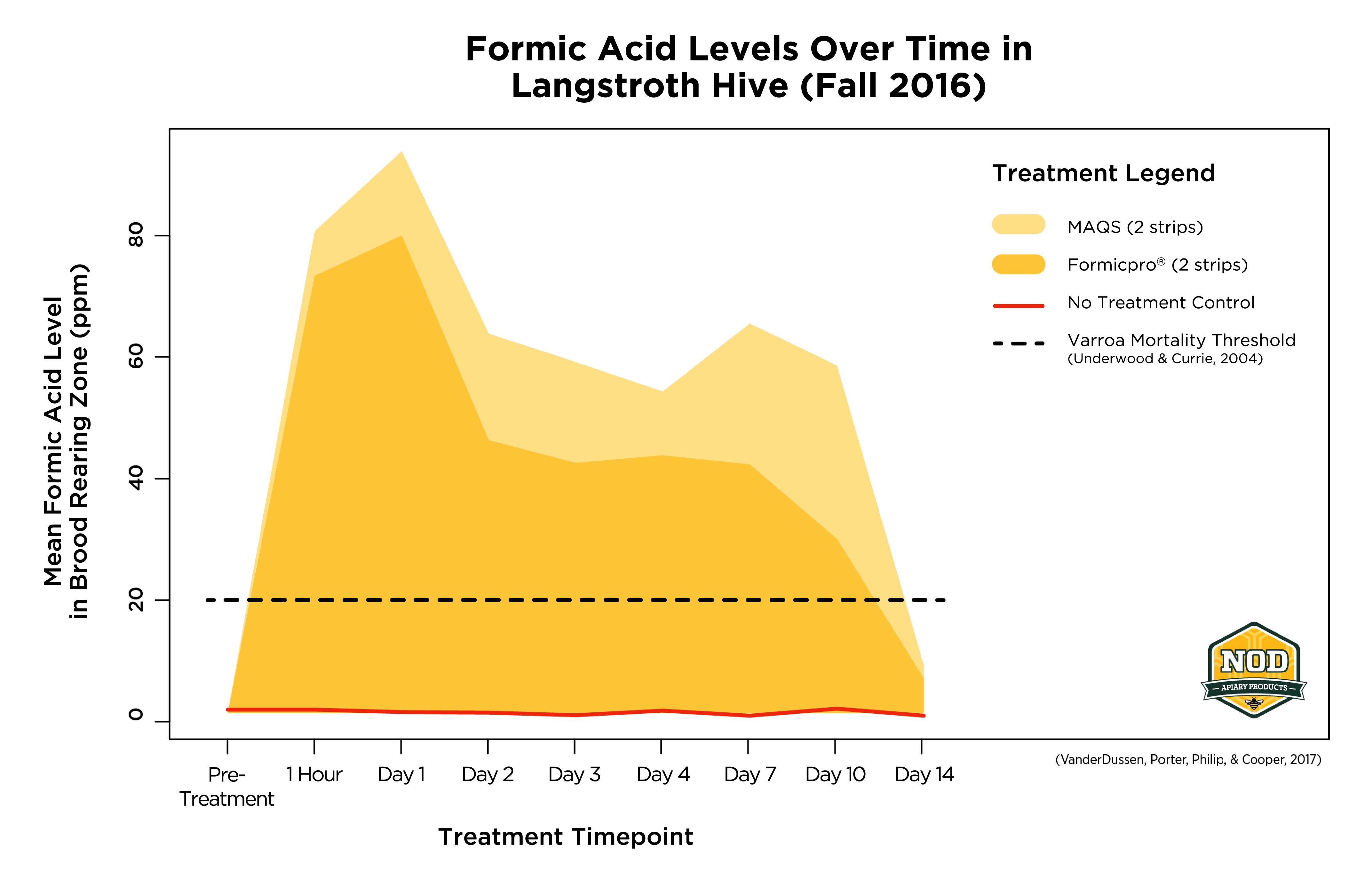
FIGURE CAPTION: Two strips of either MAQS or Formic Pro raise the level of formic acid inside the colony enough to provide excellent varroa control.
IMAGE CREDIT: Heather Broccard-Bell and Laura Wagdin.
MAQS and Formic Pro: Harnessing the Power of Formic Acid
Formic acid evaporates easily, readily moves around inside the hive, and the excess is flushed out by the bees through fanning. Beekeepers and scientists have determined the dose of formic acid that effectively controls varroa while leaving the colony unharmed. With the help of the compostable saccharide matrix and Ecopaper of MAQS and Formic Pro, beekeepers can now easily harness the power of formic acid inside their honey bee colonies to obtain optimal varroa control.
Footnotes
- It is often said that the dose makes the poison. That is, for every substance, there exists a dose small enough to not be harmful, and a dose large enough to cause damage. Even essential substances, such as water and oxygen, are deadly at high enough doses. Fortunately for us – and the field of medicine — it is also the case that often, what is a deadly dose of a given substance for one organism leaves other types of organisms unscathed. This is the principal underlying antibiotics, which kill disease-causing microorganisms while sparing host cells.But drink enough penicillin, and you too will experience penicillin toxicity.
- Like all products, MAQS and Formic Pro are designed to work under a specific set of conditions. To obtain optimal results, be sure to precisely follow the label directions for application.
About Heather Broccard-Bell, Ph.D.
Dr. Heather Broccard-Bell is the Honey Bee Health Researcher at NOD Apiary Products. She is a scientist and educator with over 15 years research and teaching experience. Heather has been focused on investigating issues surrounding honey bee health and communication since 2014. When Heather’s not in the lab, you can usually find her in the bee yard or on a trail hiking with her many pawed pals. If you’d like to ask Dr. Heather Broccard-Bell more about her work or this article, you can email her at heatherb@nodglobal.com





